![PDF] From absolute potentials to a generalized computational standard hydrogen electrode for aqueous and non-aqueous solvents. | Semantic Scholar PDF] From absolute potentials to a generalized computational standard hydrogen electrode for aqueous and non-aqueous solvents. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/57137ca4f340de92194b452bc06e730433d054d6/8-Figure4-1.png)
PDF] From absolute potentials to a generalized computational standard hydrogen electrode for aqueous and non-aqueous solvents. | Semantic Scholar

organic chemistry - How does the polarity of the eluent and sample affect the Rf value in thin layer chromatography? - Chemistry Stack Exchange

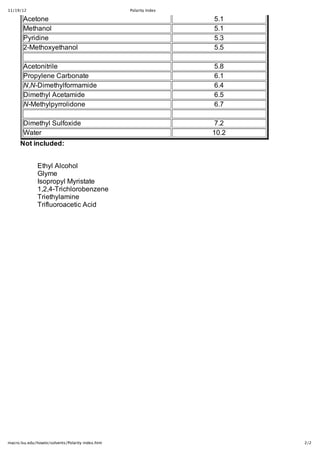
Solvent Polarity and Organic Reactivity in Mixed Solvents: Evidence Using a Reactive Molecular Probe To Assess the Role of Preferential Solvation in Aqueous Alcohols | The Journal of Organic Chemistry
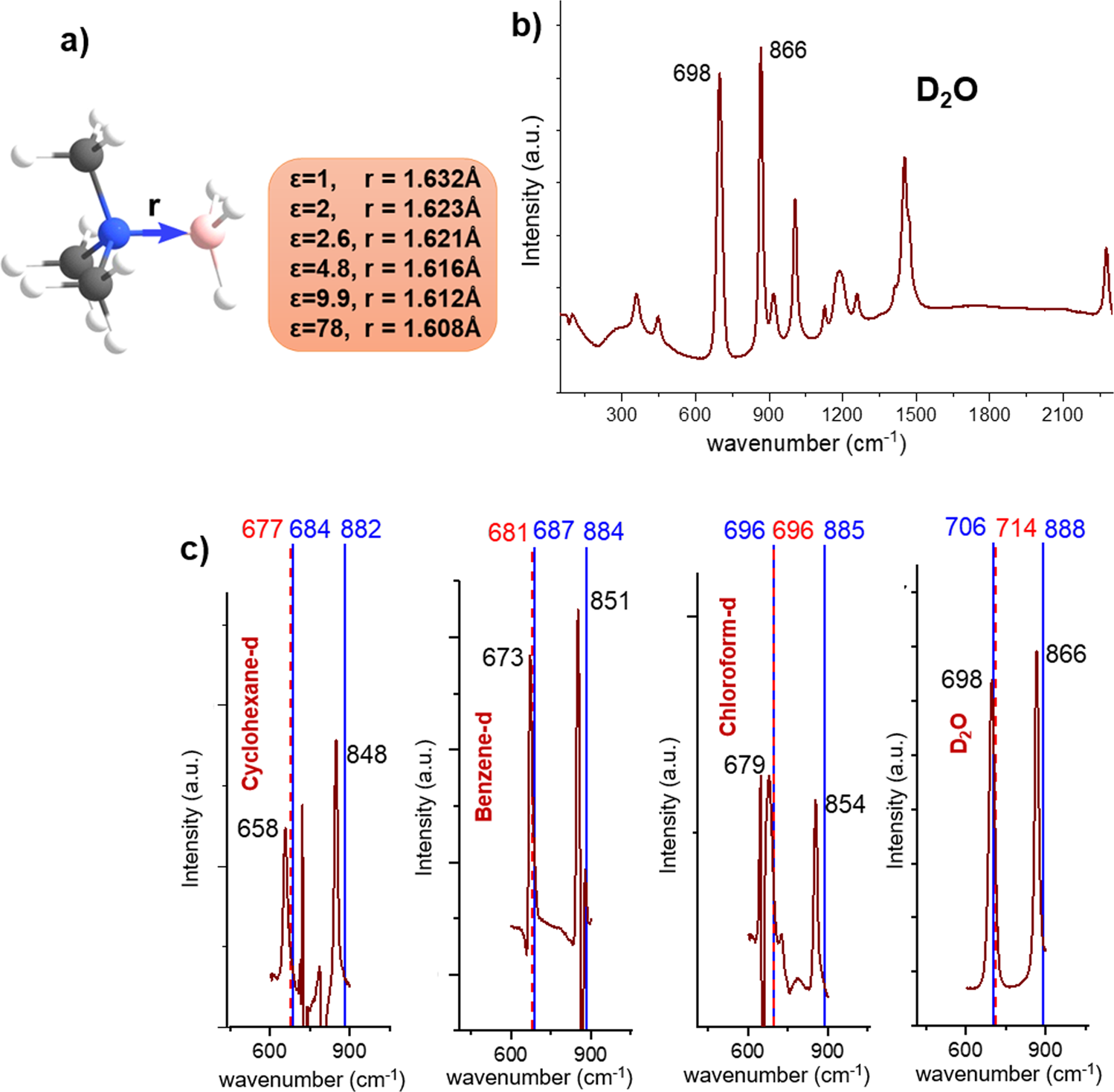
The stability of covalent dative bond significantly increases with increasing solvent polarity | Nature Communications

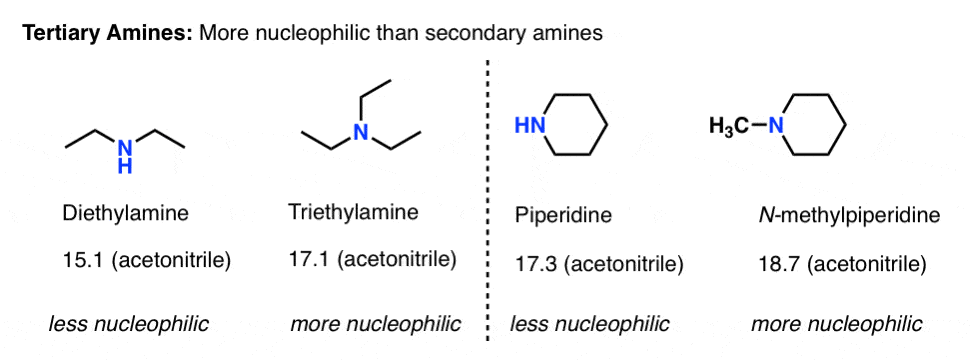
Explain why triethylamine, (CH_3CH_2)_3N, is very slightly soluble in water but readily soluble (with stirring) in 1.5 M HCl. | Homework.Study.com
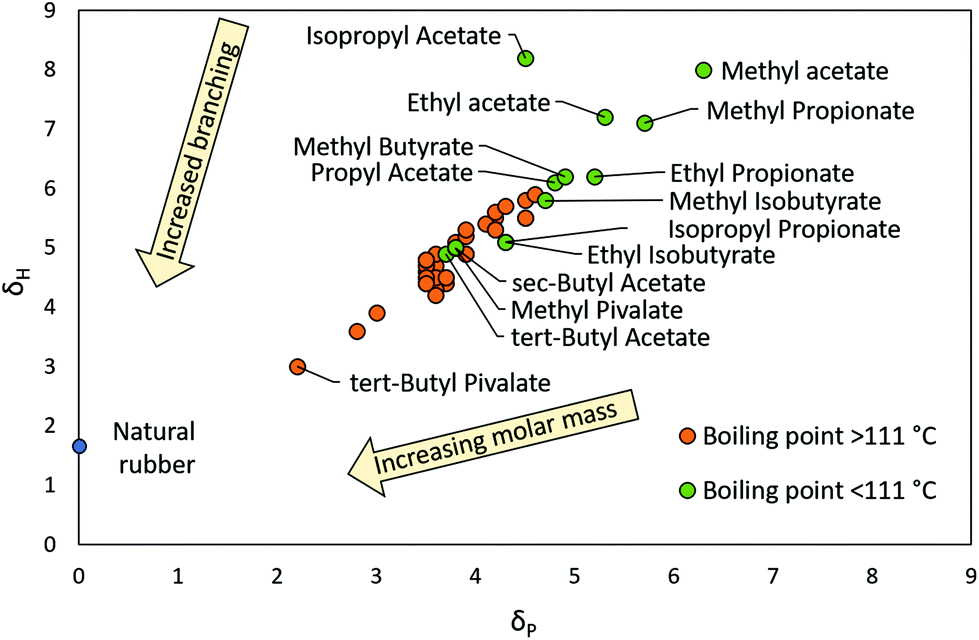
A methodical selection process for the development of ketones and esters as bio-based replacements for traditional hydrocarbon solvents - Green Chemistry (RSC Publishing) DOI:10.1039/C8GC01132J

Solvent-driven aqueous separations for hypersaline brine concentration and resource recovery: Trends in Chemistry