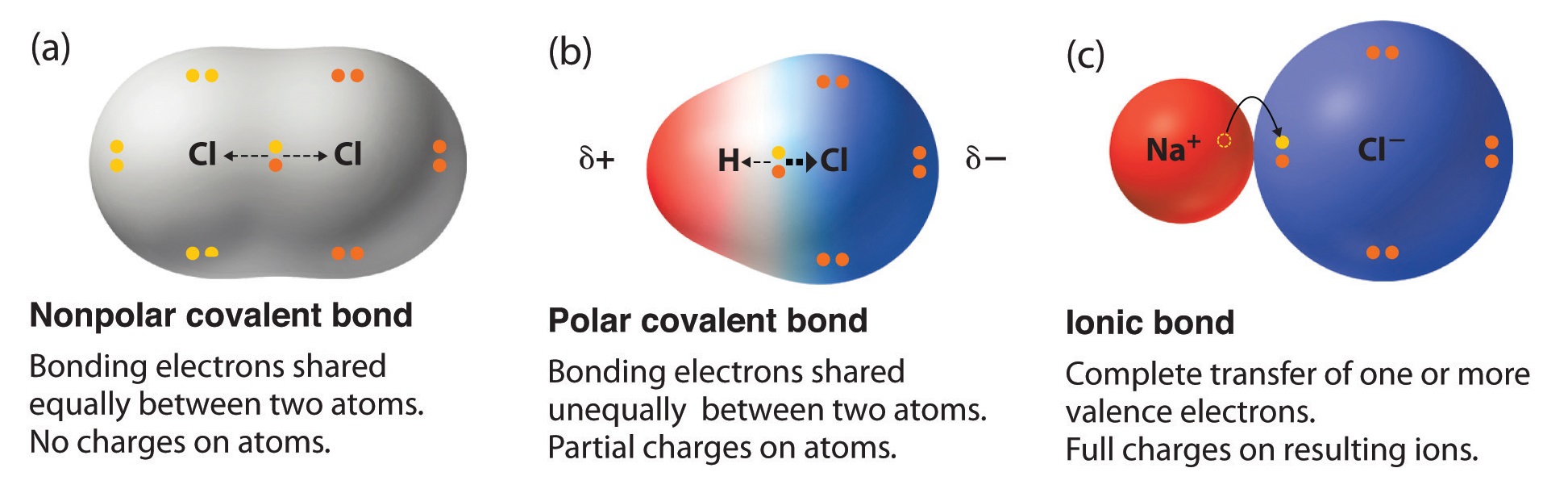
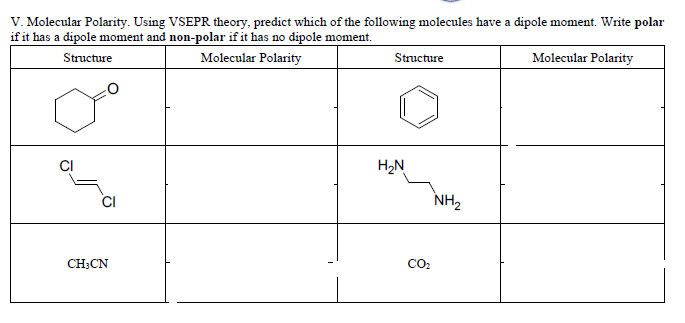
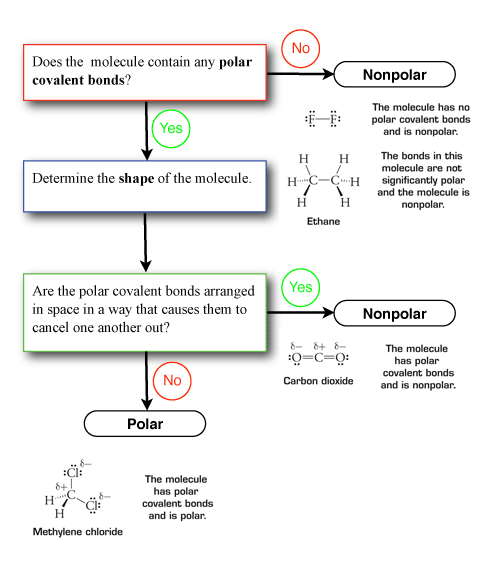
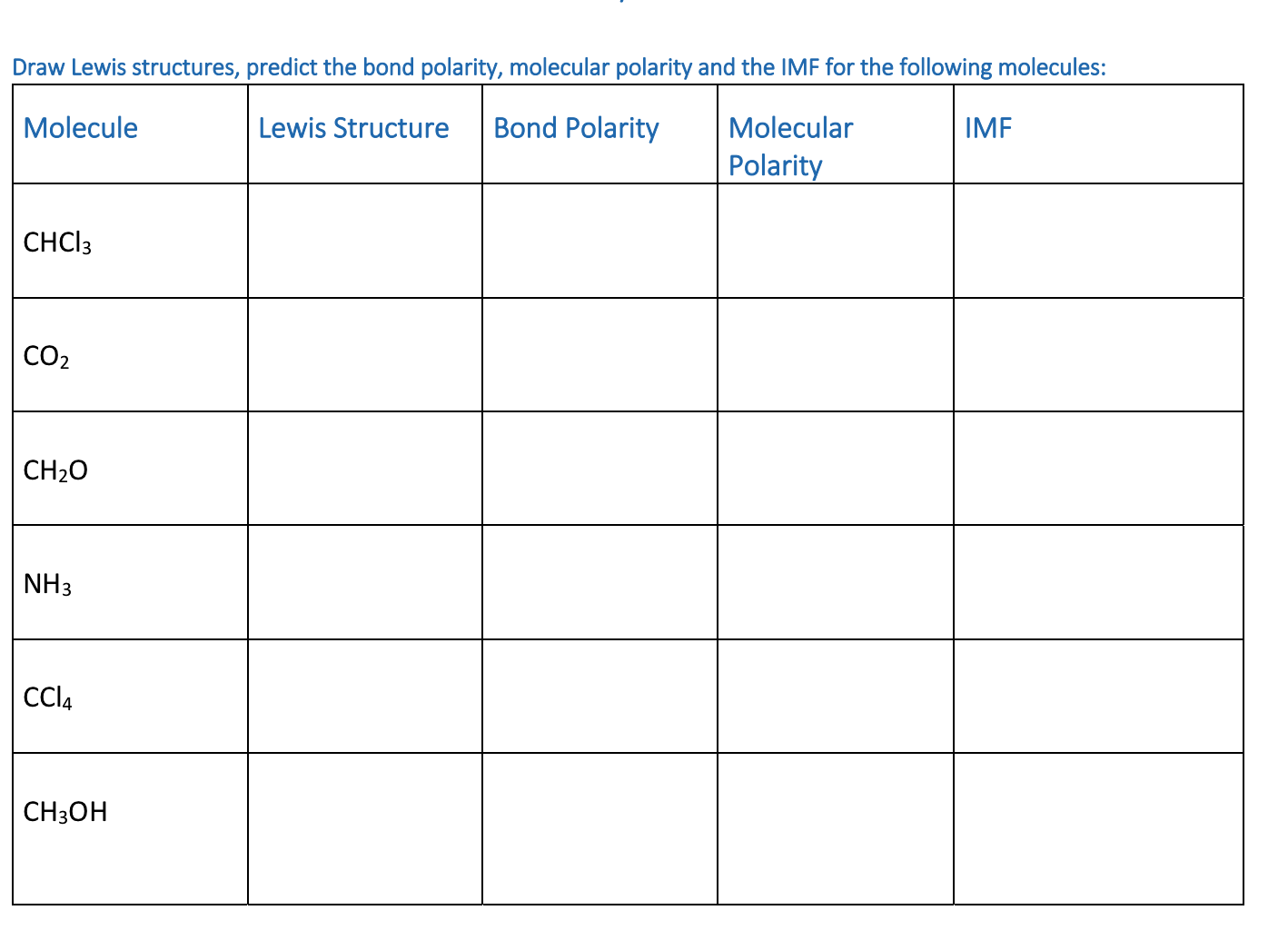
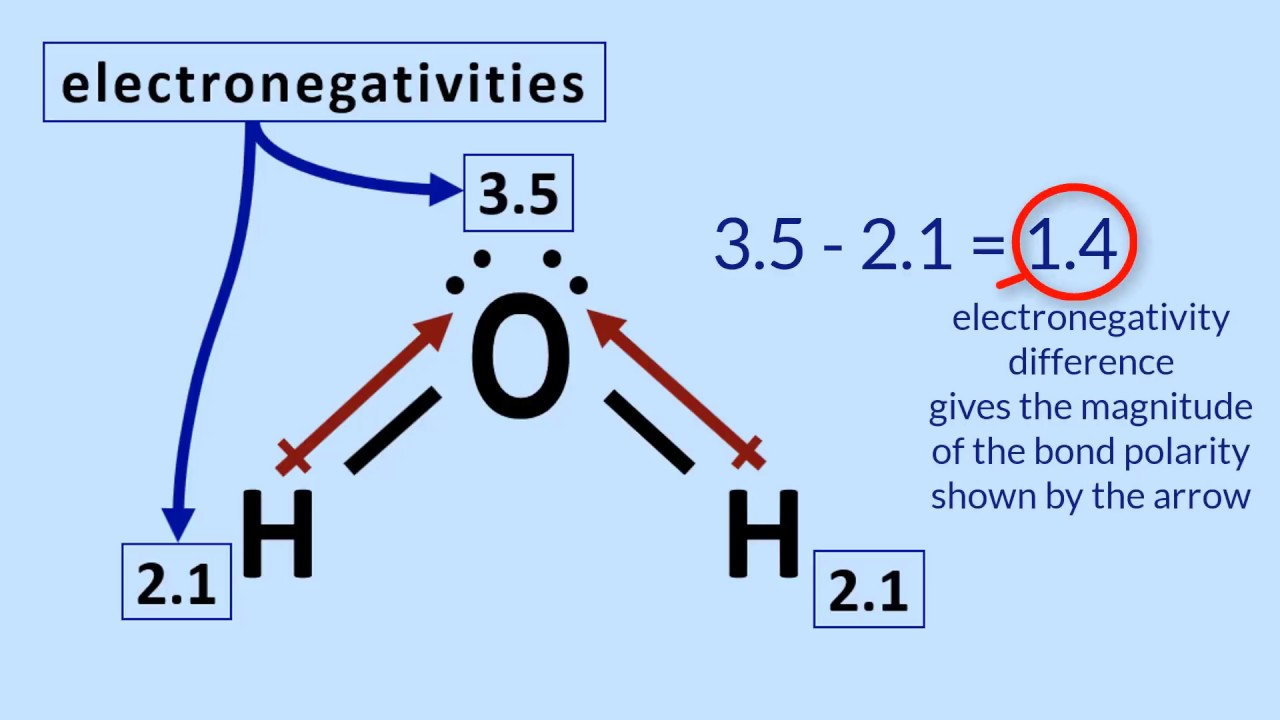
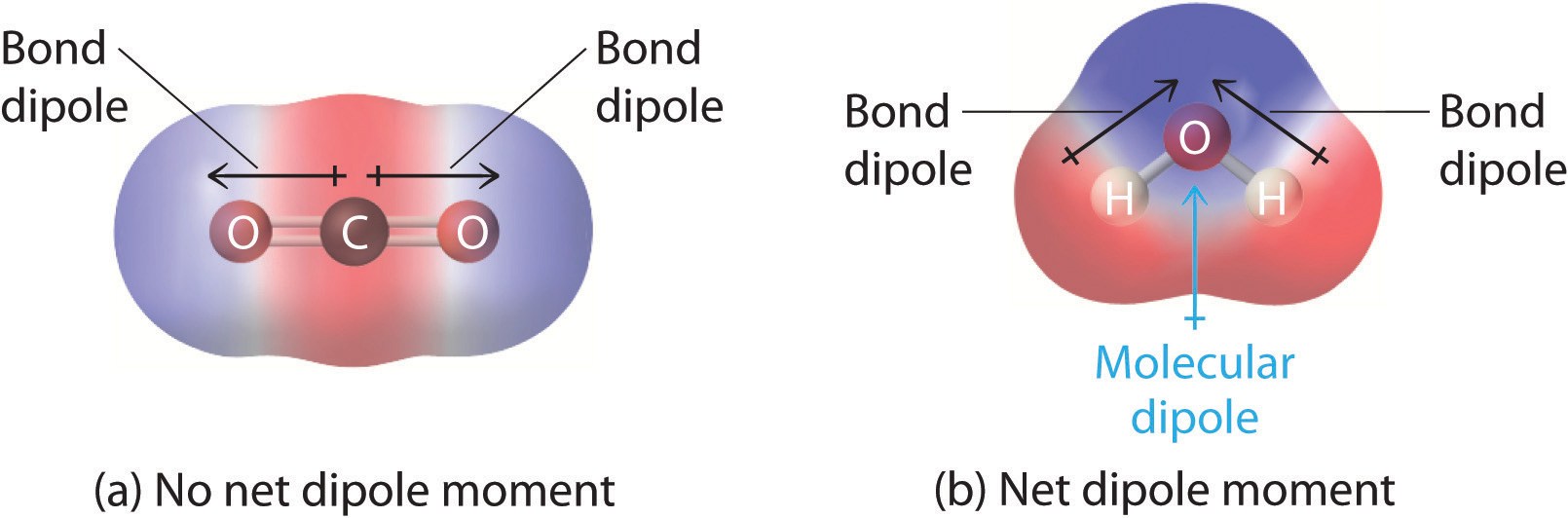
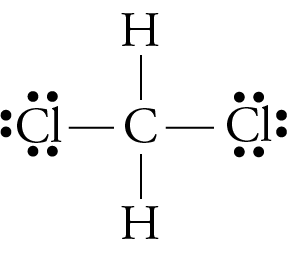
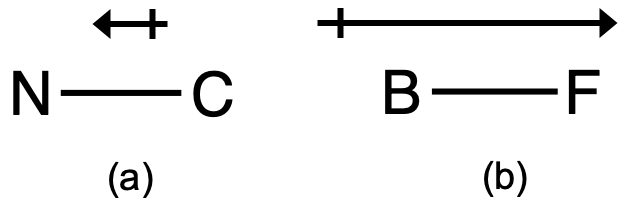Predicting Molecular Polarity When there are no polar bonds in a molecule, there is no permanent charge difference between one part of the molecule and. - ppt download
![SOLVED: Question 1 4 pts For each of the following, use the molecular shape and electronegativity (EN) values and trends to predict the molecular polarity: 1 Select ] PF3 2 [Select ] SOLVED: Question 1 4 pts For each of the following, use the molecular shape and electronegativity (EN) values and trends to predict the molecular polarity: 1 Select ] PF3 2 [Select ]](https://cdn.numerade.com/ask_images/e7e6708c6a544ab8b7e7c590c98819a5.jpg)
SOLVED: Question 1 4 pts For each of the following, use the molecular shape and electronegativity (EN) values and trends to predict the molecular polarity: 1 Select ] PF3 2 [Select ]