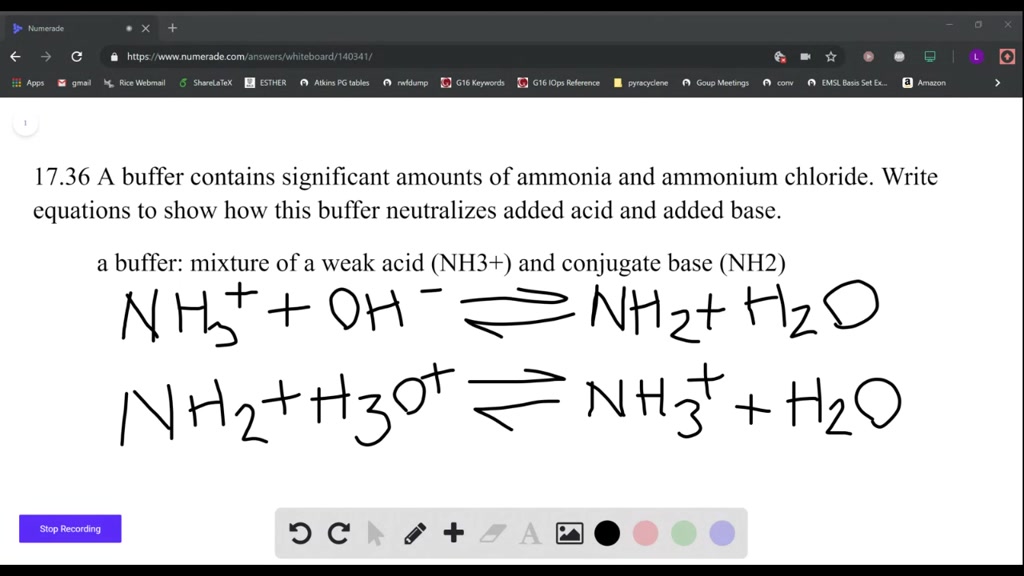
a buffer solution contain NH3 and NH4CL total concentration of buffering agent is 0 6 molar if the pOH of - Chemistry - Equilibrium - 14195039 | Meritnation.com

A buffer is generated using NH3 and NH4Cl Which of the following is/are true I Any - Chemistry - Equilibrium - 12558579 | Meritnation.com

A buffer solution made with NH3 and NH4Cl has a pH of 10.0 which procedure could be used to lower the pH ? 1.Adding HCl , 2.Adding NH3 , 3. Adding NH4Cl
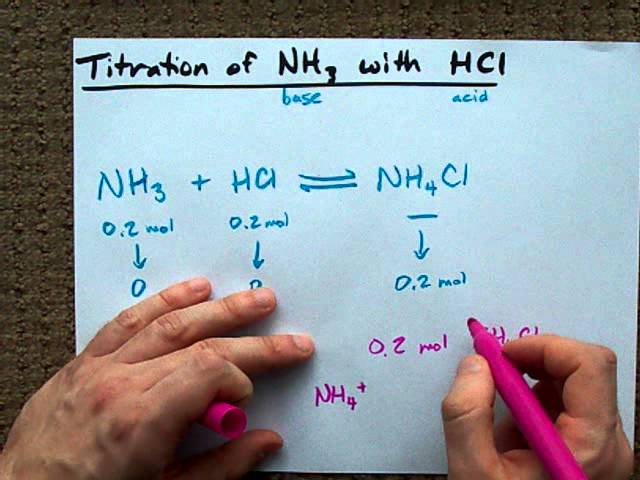
Calculate the amount of NH3 and NH4Cl required to prepare a buffer solution of pH = 9 when total concentration of buffering - Sarthaks eConnect | Largest Online Education Community

A buffer solution containing NH3 and NH4Cl has a pH value of 9. pKb for NH3 is 4.7. If in the buffer solution total concentration of buffering reagents is 0.6 mol L^(-1),