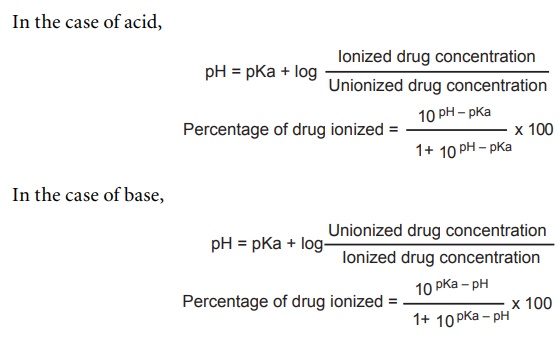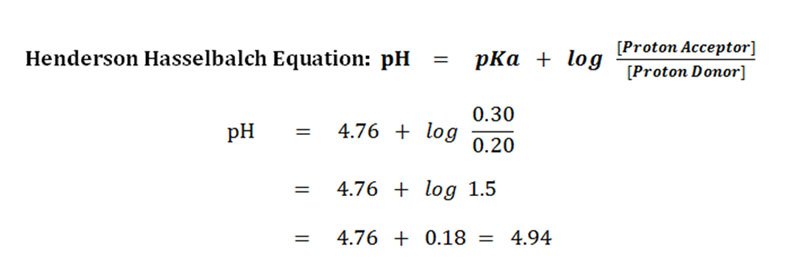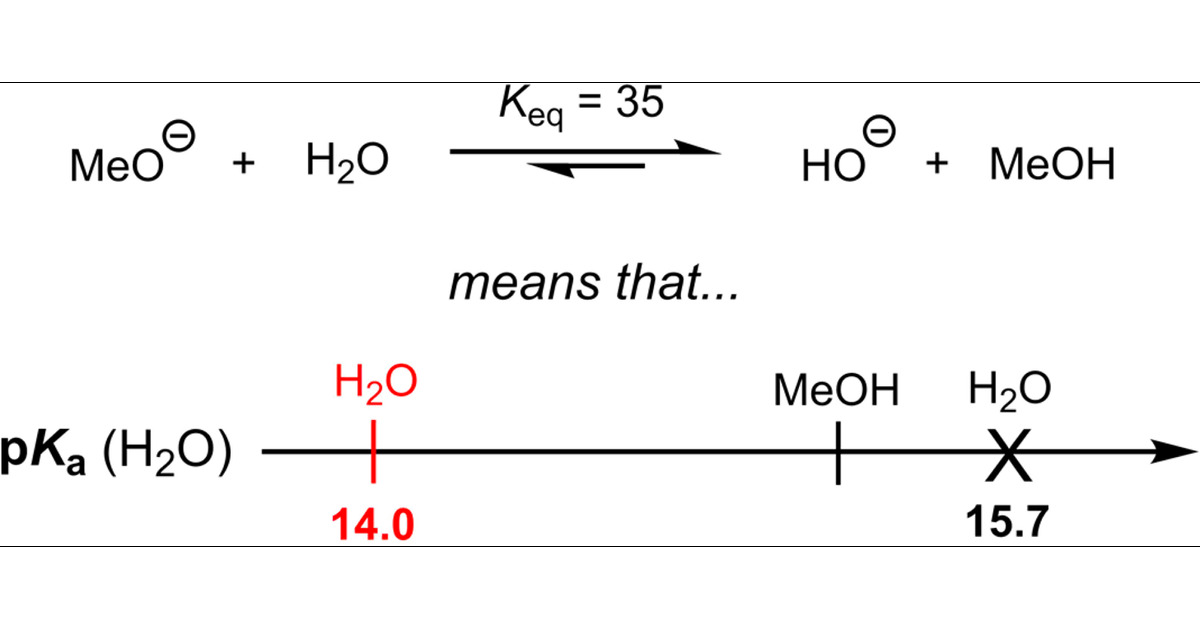
pKa Values in the Undergraduate Curriculum: What Is the Real pKa of Water? | Journal of Chemical Education

pH, pOH, H3O+, OH-, Kw, Ka, Kb, pKa, and pKb Basic Calculations -Acids and Bases Chemistry Problems - YouTube
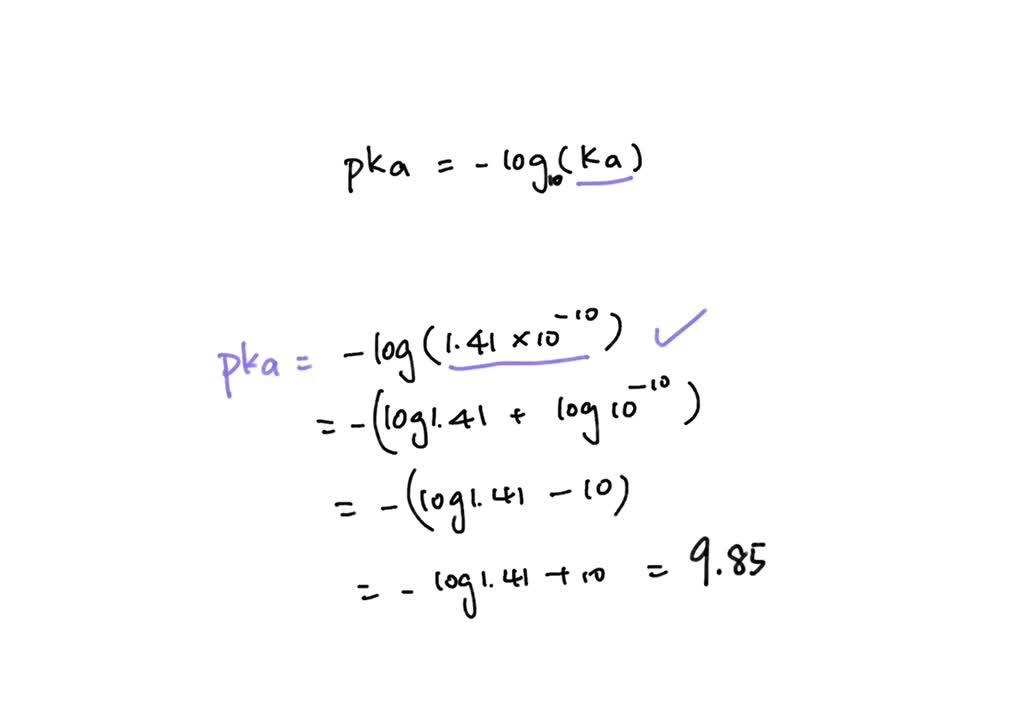
SOLVED: How would you calculate the pka or ka for CH2ClCH2COOH and CH2FCH2COOH if you only knew that this is a carboxylic acid? Do you write the conjugate acid and base to









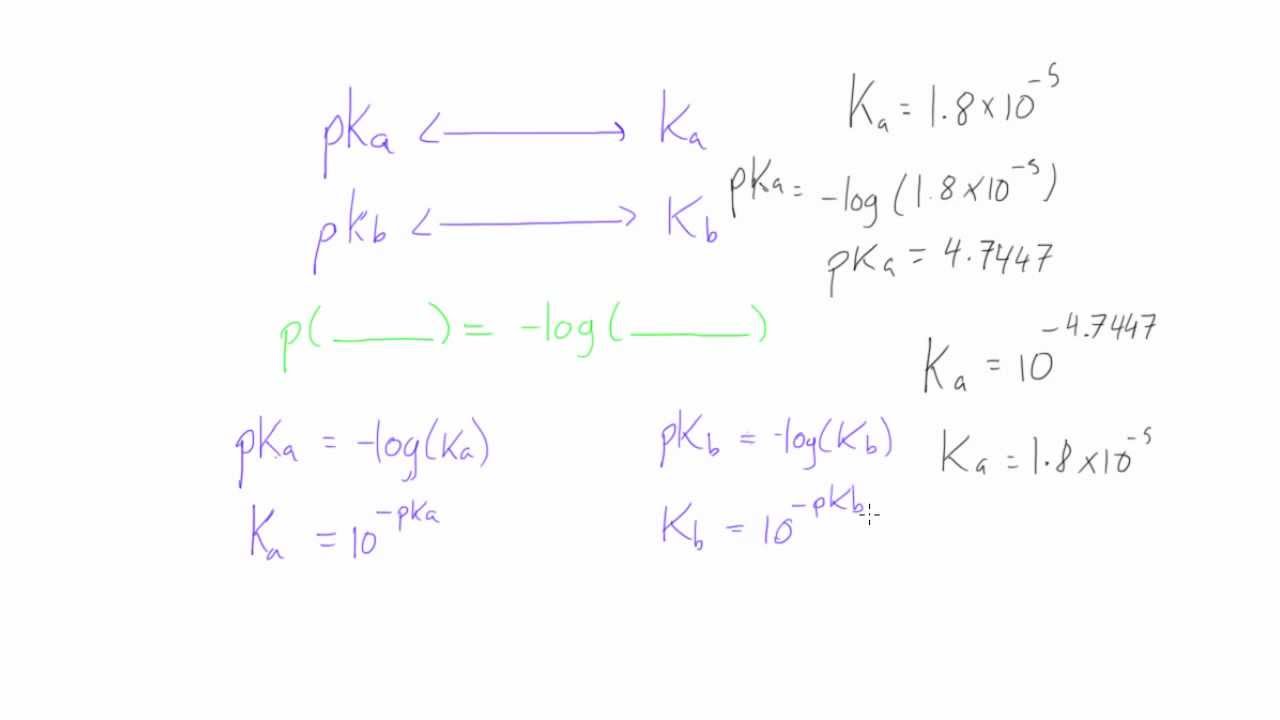









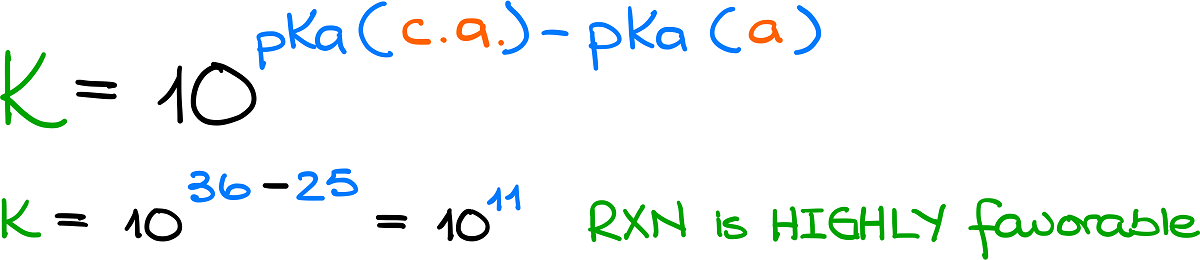
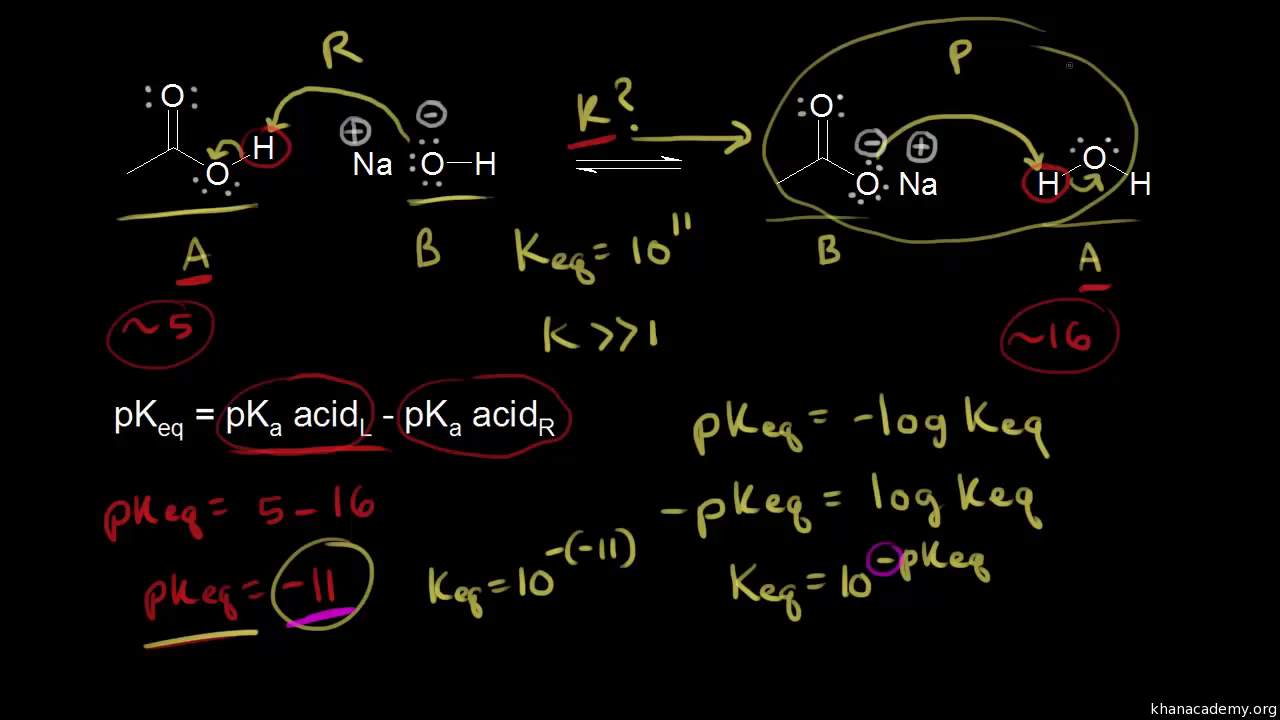
:max_bytes(150000):strip_icc()/what-is-pka-in-chemistry-605521_FINAL2-9fdfc39e9aa34caa96d6e74a2c687707.png)