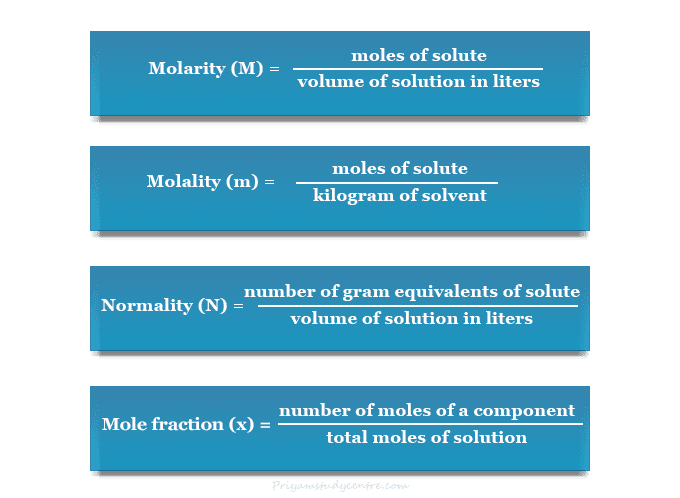
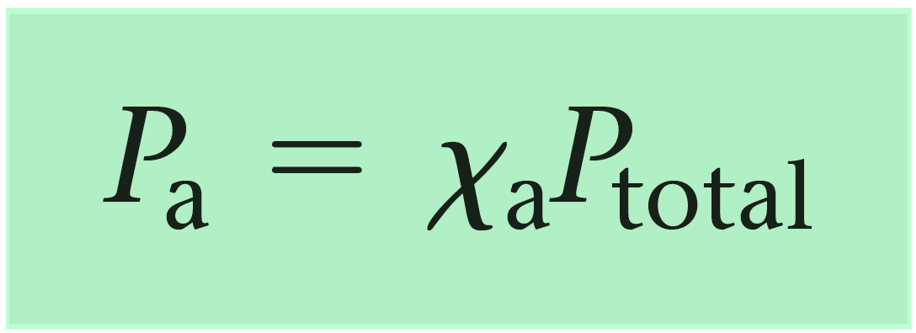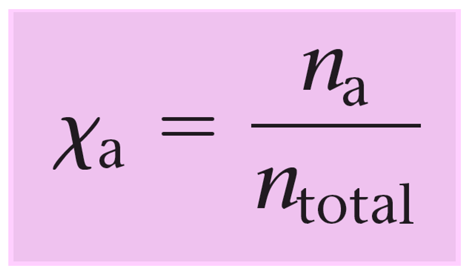![Calculate the mole fraction of water in a sodium hydroxide solution which has 80 g of NaOH and 54 g of H2O. [Relative atomic masses : Na = 23, O = 16, H = 1]. - Zigya Calculate the mole fraction of water in a sodium hydroxide solution which has 80 g of NaOH and 54 g of H2O. [Relative atomic masses : Na = 23, O = 16, H = 1]. - Zigya](https://www.zigya.com/application/zrc/images/qvar/ICHEN12115567.png)
Calculate the mole fraction of water in a sodium hydroxide solution which has 80 g of NaOH and 54 g of H2O. [Relative atomic masses : Na = 23, O = 16, H = 1]. - Zigya
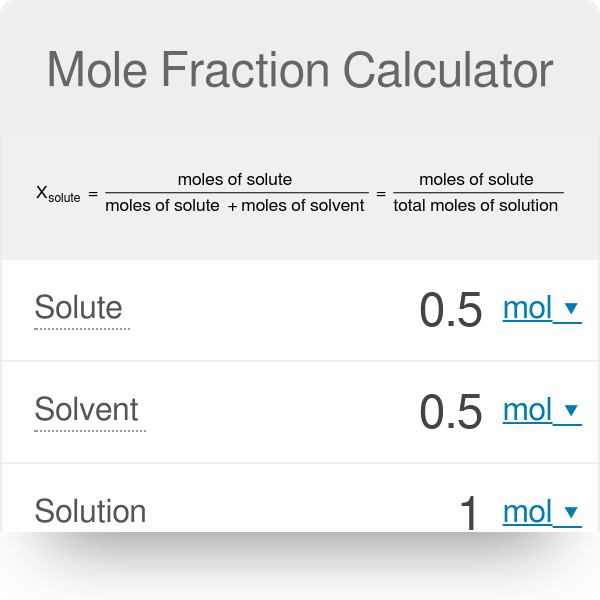
What is the mole fraction of the solute and the solvent given the 5.02g of KNO4 in 95.0 g of solvent? - Quora

SOLVED: Questions in mole fraction Calculate the mole fraction, molality and molarity of HNOz in a solution containing 12.2 % HNOz: Given density of HNO3 as 1.038 g cm 3,H= 1,N= 1+,0 =

Question Video: Determining the Mole Fraction of a Gas Given the Mole Fraction of the Other Gas in the Mixture | Nagwa