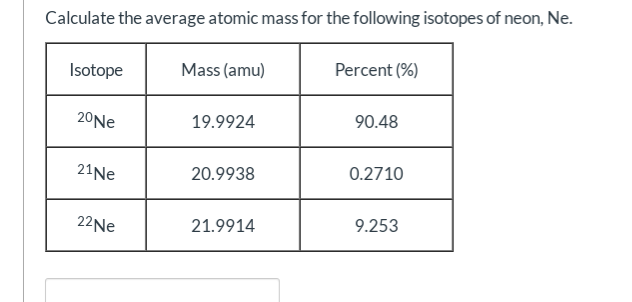
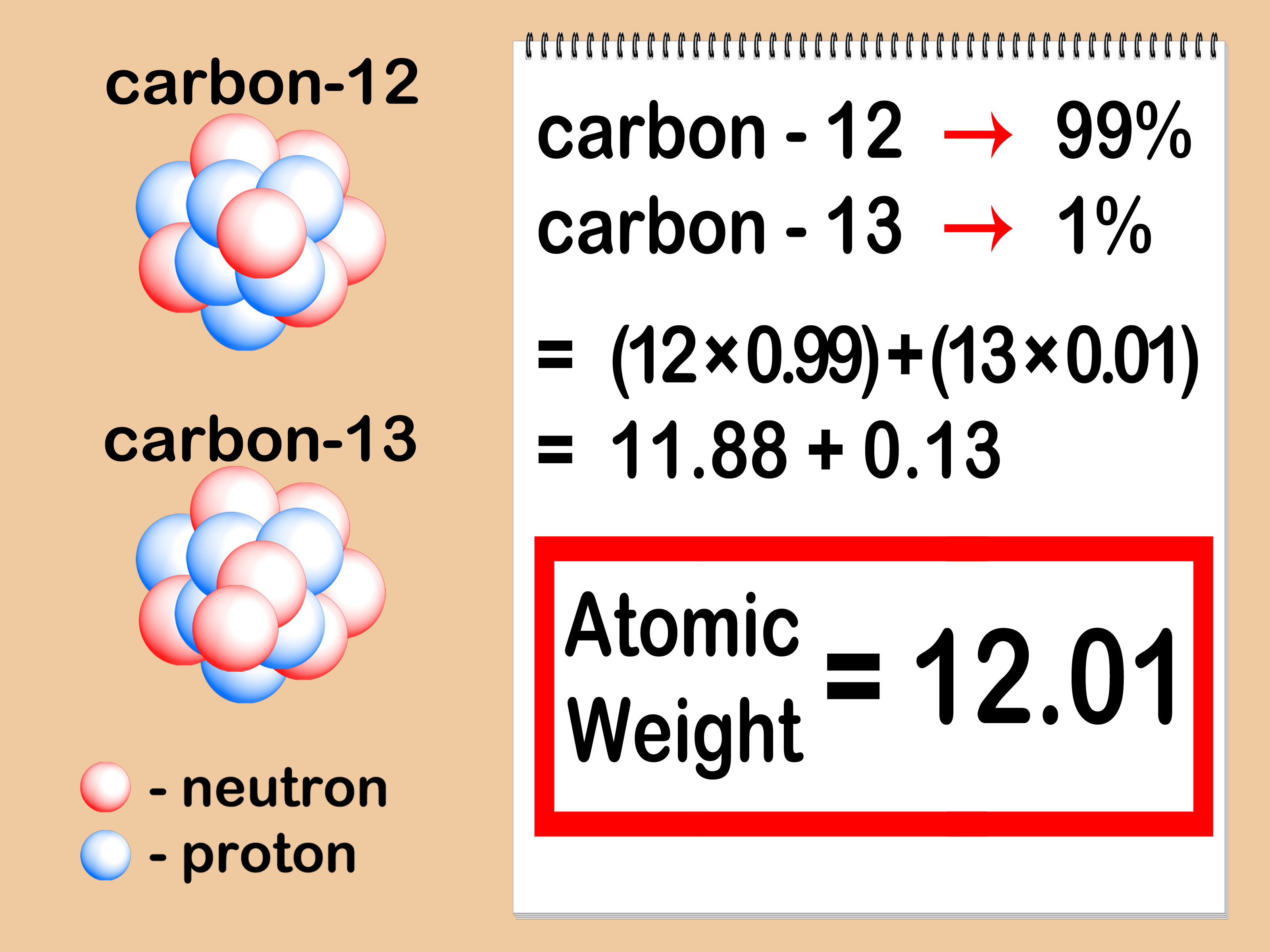
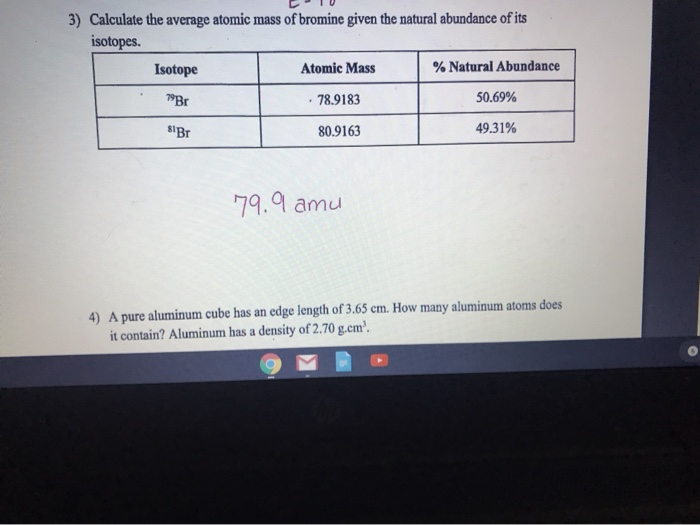
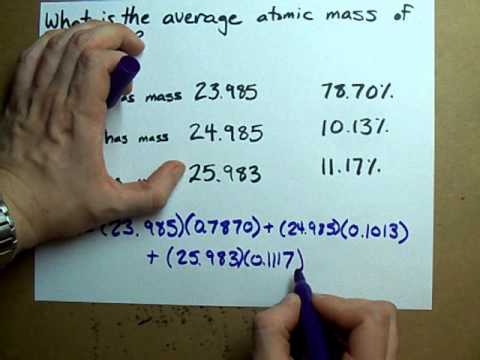Isotope &per Natural abundance &Molar mass ^1H &99.985 &1 ^2H &0.015 & 2 The average atomic mass of hydrogen using the following data is .

Average Atomic Mass In nature, most elements are a mixture of different isotopes The mass of a sample of an element is a weighted average of all the isotopes. - ppt download
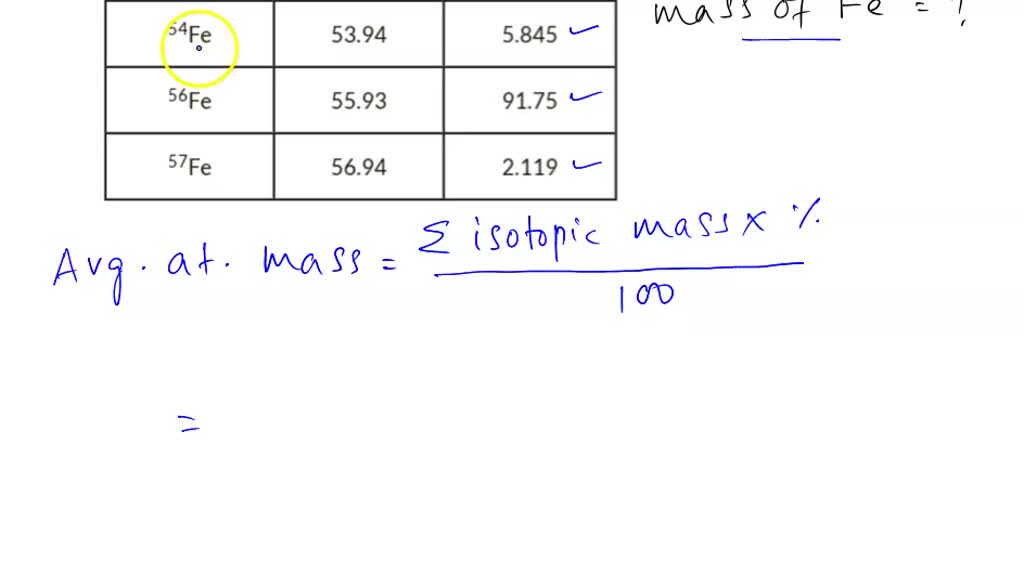
SOLVED: 'Partial calculating average atomic mass sng tne inforation in te tabl * | cOIPlayer/ he Structure of the Atom ssignment Active Practice calculating average atomic mass Using the information in the"