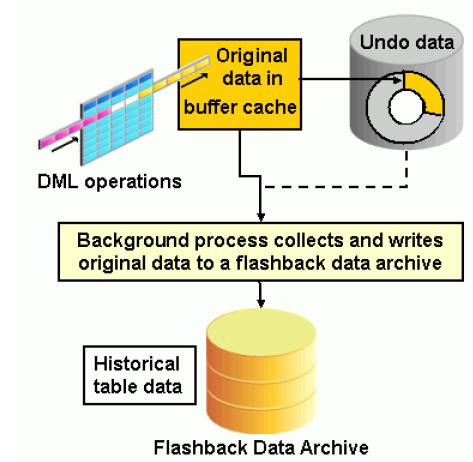
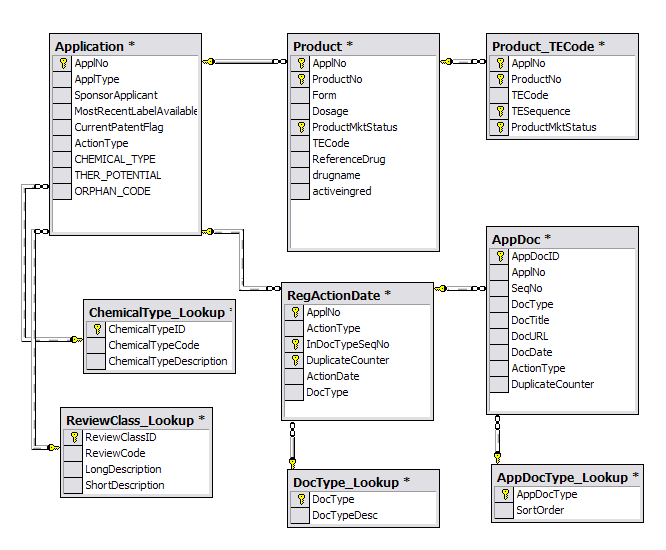
UCLA CTSI | Accelerating Discoveries Toward Better Health | NIH and FDA Announce New Template for IND and IDE Protocols
GUIDE TO PRIMARY & SECONDARY SOURCES ON THE SUBURBANIZATION OF MONTGOMERY COUNTY, MD FINDING AID US Food and Drug Administr

FDA shutters its alternative device report database, disclosing 20 years of safety data | Fierce Biotech

KCAS Webinar Archive: Impact of New FDA Guidance on Bioanalytical Testing on Drug Development | KCAS Bioanalytical Services